|
1/16/2024 0 Comments Keynote 048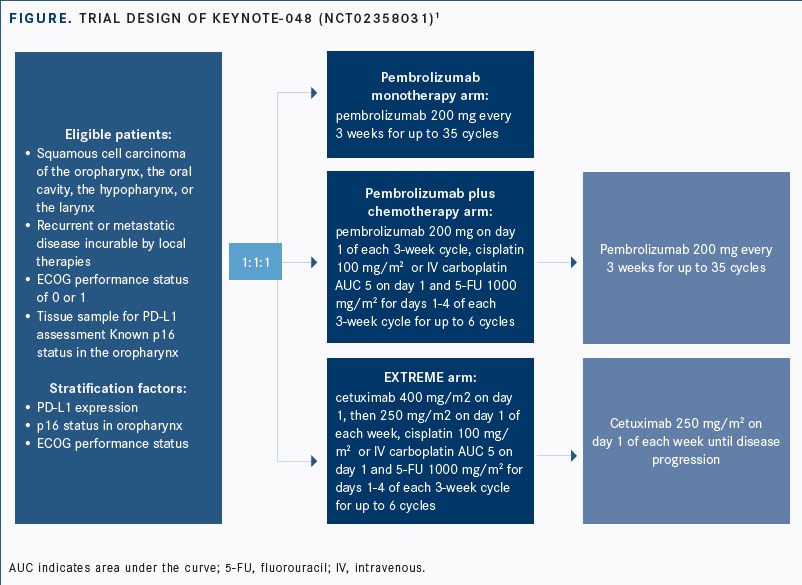 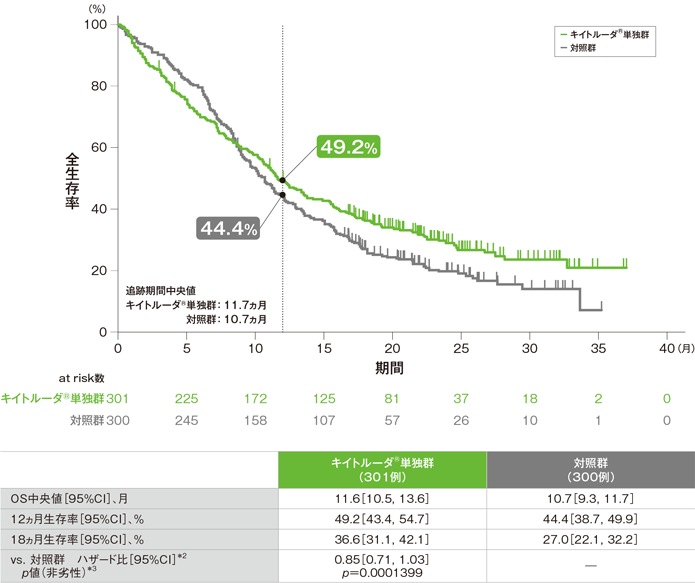
Abbreviationsĥ-FU = 5-Fluorouracil AE = Adverse Event AEOSI = Adverse Events Of Special Interest AUC 5 = Desired Carboplatin Exposure of 5 mg/ml CI = Confidence Interval CPS = Combined Positive Score CR = Complete Response DoR = Duration Of Response ECOG PS = Eastern Cooperative Oncology Group Performance Status ESMO = European Society For Medical Oncology EXTREME = Cetuximab + 5-Fluorouracil + Platinum-Based Chemotherapy HNSCC = Head And Neck Squamous Cell Carcinoma HR = Hazard Ratio IHC = Immunohistochemistry ITT = Intention To Treat IV = Intravenous mo = Month M/uR = Metastatic Or Unresectable Recurrent No. KEYNOTE-048 trial was a multicentre, randomised, open-label, active-controlled Phase III study. *The KEYNOTE-048 study employed the PD-L1 IHC 22C3 pharmDx assay (Agilent Technologies, Carpinteria, CA, USA). PD-L1 testing supports a targeted treatment approach for all appropriate patients with M/uR HNSCC* 1 CPS ≥ 1 in the KEYNOTE-048 study 1 It concluded that 400 mg Q6W dosing regimen for KEYTRUDA monotherapy and combination is predicted to have a similar efficacy and safety profile as the approved 200 mg Q3W dosing regimen. The study that led to the approval of the Q6W for monotherapy and combination patients assessed the 400 mg Q6W dosing schedule based on an exposure-response evaluation using modelling and simulation. Both doses are administered as IV infusions over 30 minutes
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
